
Improved thermodynamic parameters and helix initiation factor to predict stability of DNA duplexes. DNA polymerase delta holoenzyme: action on single-stranded DNA and on double-stranded DNA in the presence of replicative DNA helicases.  
Replication dynamics of the yeast genome. RNase H2-initiated ribonucleotide excision repair. Exonucleolytic proofreading by calf thymus DNA polymerase delta. Abundant ribonucleotide incorporation into DNA by yeast replicative polymerases. DNA polymerase delta is highly processive with proliferating cell nuclear antigen and undergoes collision release upon completing DNA. Formation and activity of complexes with the proliferating cell nuclear antigen and with DNA polymerases delta and epsilon. Saccharomyces cerevisiae replication factor C. Role of induced fit in enzyme specificity: a molecular forward/reverse switch. Distribution of functions between FEN1 AND DNA2. Pre-steady state kinetic studies of the fidelity of nucleotide incorporation by yeast DNA polymerase delta. Sequential switching of binding partners on PCNA during in vitro Okazaki fragment maturation. Coordination of multiple enzyme activities by a single PCNA in archaeal Okazaki fragment maturation. Polymerase exchange on single DNA molecules reveals processivity clamp control of translesion synthesis. A sliding-clamp toolbelt binds high- and low-fidelity DNA polymerases simultaneously. Indiani, C., McInerney, P., Georgescu, R., Goodman, M.F. The 3′→5′ exonuclease of DNA polymerase delta can substitute for the 5′ flap endonuclease Rad27/Fen1 in processing Okazaki fragments and preventing genome instability. Dna2 on the road to Okazaki fragment processing and genome stability in eukaryotes. Idling by DNA polymerase delta maintains a ligatable nick during lagging-strand DNA replication. Garg, P., Stith, C.M., Sabouri, N., Johansson, E. Flexibility of eukaryotic Okazaki fragment maturation through regulated strand displacement synthesis. Stith, C.M., Sterling, J., Resnick, M.A., Gordenin, D.A. Bacteriophage T4 RNase H removes both RNA primers and adjacent DNA from the 5′ end of lagging strand fragments. The eukaryotic leading and lagging strand DNA polymerases are loaded onto primer-ends via separate mechanisms but have comparable processivity in the presence of PCNA. Functions of replication factor C and proliferating-cell nuclear antigen: functional similarity of DNA polymerase accessory proteins from human cells and bacteriophage T4. PCNA binding domains in all three subunits of yeast DNA polymerase δ modulate its function in DNA replication. Eukaryotic DNA polymerases require an iron-sulfur cluster for the formation of active complexes. Direct interaction of proliferating cell nuclear antigen with the small subunit of DNA polymerase delta. 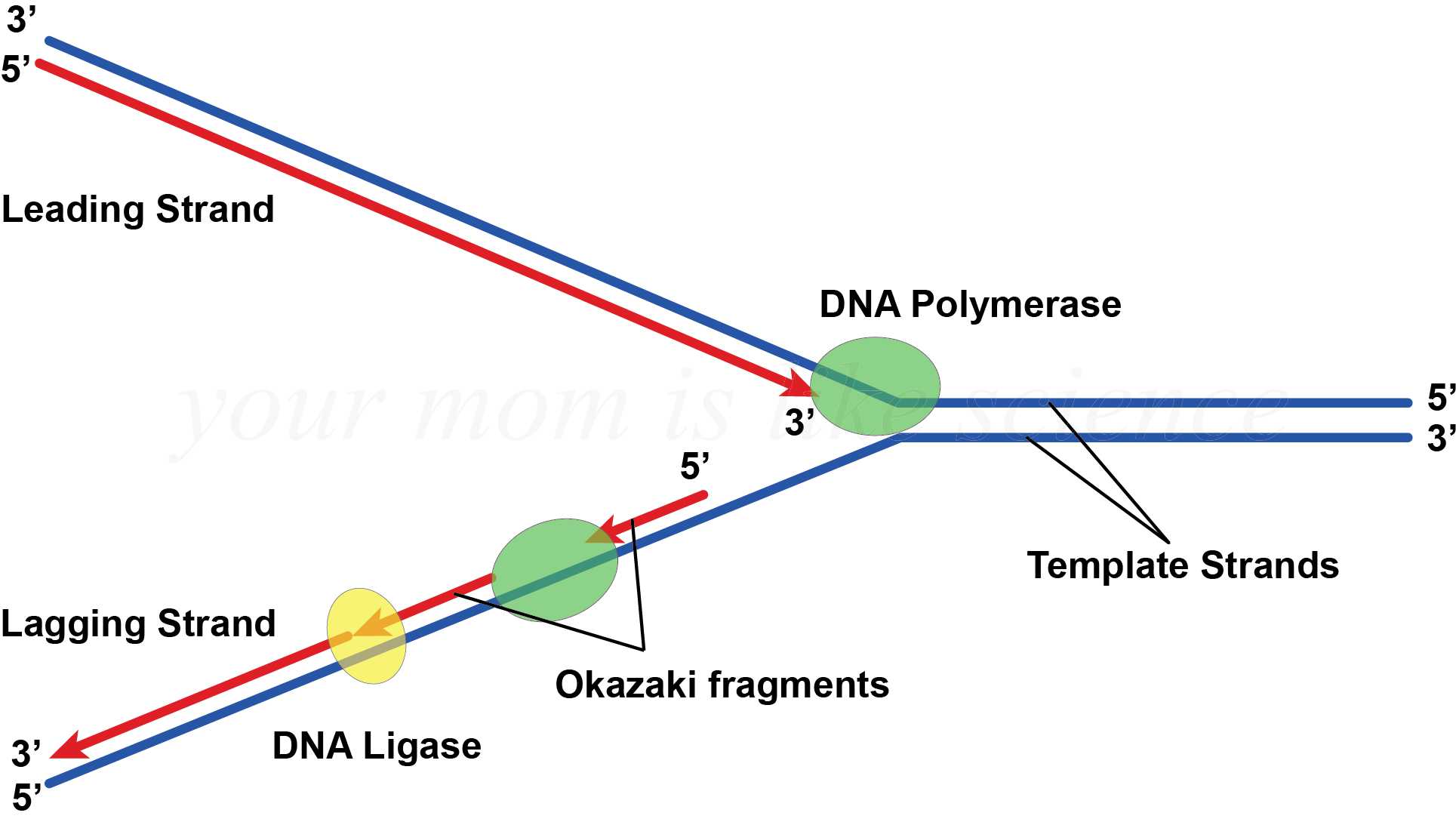
The influence of the Cdc27 subunit on the properties of the Schizosaccharomyces pombe DNA polymerase delta. Characterization of the two small subunits of Saccharomyces cerevisiae DNA polymerase delta. Lagging-strand replication shapes the mutational landscape of the genome. Eukaryotic lagging strand DNA replication employs a multi-pathway mechanism that protects genome integrity. Mechanism for priming DNA synthesis by yeast DNA polymerase α. Finally, whereas elongation rates are under dynamic dNTP control, maturation rates are buffered against changes in dNTP concentrations. Kinetic and enzyme-trapping experiments support a model in which a stable PCNA–DNA–Pol δ–FEN1 complex moves processively through iterative steps of nick translation, ultimately completely removing primer RNA. Thus, the nascent flap acts as a progressive molecular brake on the polymerase, and consequently FEN1 cuts predominantly single-nucleotide flaps. When Pol δ invaded double-stranded RNA–DNA representing unmatured Okazaki fragments, the incorporation rate of each nucleotide decreased successively to 10–20% that of the preceding nucleotide. During elongation, PCNA increased the Pol δ catalytic rate by >30-fold. We studied this process in the Saccharomyces cerevisiae machinery at millisecond resolution. Pol δ and the flap endonuclease FEN1, coordinated by the PCNA clamp, remove RNA primers and produce ligatable nicks. DNA polymerase delta (Pol δ) is responsible for elongation and maturation of Okazaki fragments.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
